Phenacaine
Jump to navigation
Jump to search

| |
| Names | |
|---|---|
| IUPAC name
(1E)-N,N′-Bis(4-ethoxyphenyl)ethanimidamide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C18H22N2O2 | |
| Molar mass | 298.386 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenacaine, also known as holocaine,[1] is a local anesthetic. It is approved for ophthalmic use.[2]
Synthesis
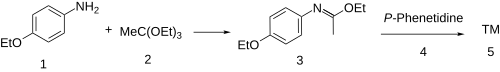
Synthesis:[3]
The condensation of P-Phenetidine (1) with triethyl orthoacetate [78-39-7] (2) to afford the imino ether (a so-called Pinner salt) (3). Reaction of that intermediate with a second equivalent of the aniline results (4) in a net displacement of ethanol, probably by an addition-elimination scheme. There is thus obtained the amidine, phenacaine (5).
In the patented synthesis,[4] phenacetin was used as precursor. Treatment with PCl3 gave the enol chloride, and reaction of this intermediate with p-phenetidine then completed the synthesis of phenacaine.
References
- ^ "Holocaine Hydrochloride".
- ^ Merck Index, 1985
- ^ DeWOLFE, ROBERT H. (1962). "Reactions of Aromatic Amines with Aliphatic Ortho Esters. A Convenient Synthesis of Alkyl N-Arylimidic Esters". The Journal of Organic Chemistry. 27 (2): 490–493. doi:10.1021/jo01049a036.
- ^ Dr Ernst Taeuber, DE 79868 (1894).
Categories:
- Articles without KEGG source
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description with empty Wikidata description
- Local anesthetics
- Phenol ethers
- Anilines
- Amidines
- All stub articles
- Nervous system drug stubs