Mesitol
Jump to navigation
Jump to search
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,4,6-Trimethylphenol | |
| Other names
Hydroxymesitylene; Mesityl alcohol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H12O | |
| Molar mass | 136.194 g·mol−1 |
| Appearance | white solid |
| Melting point | 70–72 °C (158–162 °F; 343–345 K)[1] |
| Boiling point | 220 °C (428 °F; 493 K)[1] |
| 1.01 g/l | |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H314, H411 | |
| P260, P264, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
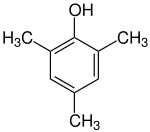
Mesitol (2,4,6-trimethylphenol) is an organic compound with the formula (CH3)3C6H2OH. It is one of several isomers of trimethylphenol. The name and structure of mesitol derives from the combination of mesitylene and phenol.
Synthesis
Mesitol is the main product from the methylation of phenol with methanol in the presence of a solid acid.[2]
It can also be obtained by reaction of mesitylene with peroxymonophosphoric acid:[3]
An alternative route involves palladium-catalyzed reaction of bromomesitylene with potassium hydroxide.[4]
References
- ^ a b "2,4,6-Trimethylphenol". Sigma-Aldrich.
- ^ Fiege, Helmut; Voges, Heinz-Werner; Hamamoto, Toshikazu; Umemura, Sumio; Iwata, Tadao; Miki, Hisaya; Fujita, Yasuhiro; Buysch, Hans-Josef; Garbe (2000). "Phenol Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313.
- ^ Ogata, Yoshiro; Sawaki, Yasuhiko; Tomizawa, Kohtaro; Ohno, Takashi (1981). "Aromatic hydroxylation with peroxymonophosphoric acid". Tetrahedron. 37 (8): 1485. doi:10.1016/S0040-4020(01)92087-3.
- ^ Anderson, Kevin W.; Ikawa, Takashi; Tundel, Rachel E.; Buchwald, Stephen L. (2006). "The Selective Reaction of Aryl Halides with KOH: Synthesis of Phenols, Aromatic Ethers, and Benzofurans". Journal of the American Chemical Society. 128 (33): 10694–10695. doi:10.1021/ja0639719. PMID 16910660.
Categories:
- Pages with short description
- Articles without InChI source
- Articles without EBI source
- Articles without KEGG source
- Pages using collapsible list with both background and text-align in titlestyle
- Chembox having GHS data
- GHS warnings
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description with empty Wikidata description
- All stub articles
- Aromatic compound stubs
- Alkylphenols
