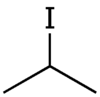
Isopropyl iodide
Jump to navigation
Jump to search
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Iodopropane[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| 1098244 | |
| ChemSpider | |
| EC Number |
|
| MeSH | isopropyl+iodide |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2392 |
| |
| |
| Properties | |
| C3H7I | |
| Molar mass | 169.993 g·mol−1 |
| Appearance | Colourless liquid |
| Density | 1.703 g mL−1 |
| Melting point | −90.00 °C; −130.00 °F; 183.15 K |
| Boiling point | 88.8 to 89.8 °C; 191.7 to 193.5 °F; 361.9 to 362.9 K |
| 1.4 g L−1 (at 12.5 °C) | |
| Solubility in chloroform | Miscible |
| Solubility in ethanol | Miscible |
| Solubility in diethyl ether | Miscible |
| Solubility in benzene | Miscible |
Henry's law
constant (kH) |
890 nmol Pa−1 kg−1 |
Refractive index (nD)
|
1.4997 |
| Viscosity | 6.971 mPa (at 20 °C) |
| Thermochemistry | |
Heat capacity (C)
|
137.3 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−77.2–−72.6 kJ mol−1 |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H226, H302 | |
| Flash point | 42 °C (108 °F; 315 K) |
| Related compounds | |
Related alkanes
|
|
Related compounds
|
Diiodohydroxypropane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isopropyl iodide is the organoiodine compound with the formula (CH3)2CHI. It is colorless, flammable, and volatile. Organic iodides are light-sensitive and take on a yellow colour upon storage, owing to the formation of iodine.
Preparation
Isopropyl iodide is prepared by iodination of isopropyl alcohol using hydrogen iodide or, equivalently, with a mixture of glycerol, iodine, and phosphorus.[2] An alternative preparation involves the reaction of 2-propyl bromide with an acetone solution of sodium iodide (Finkelstein reaction):[3]
- (CH3)2CHBr + NaI → (CH3)2CHI + NaBr
References
- ^ "isopropyl iodide - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 27 March 2005. Identification and Related Records. Retrieved 3 March 2012.
- ^ Merck Index of Chemicals and Drugs, 9th ed., monograph 5074
- ^ Textbook of Practical Organic Chemistry, 5th Edition, Prentice Hall, 1989
Categories:
- Articles without InChI source
- Articles without EBI source
- Articles without KEGG source
- Articles with changed ChemSpider identifier
- Articles with changed InChI identifier
- Pages using collapsible list with both background and text-align in titlestyle
- Chembox having GHS data
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description with empty Wikidata description
- All stub articles
- Organohalide stubs
- Iodoalkanes
- Isopropyl compounds
- Iodides