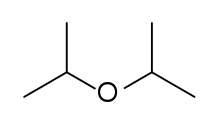
Diisopropyl ether
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-[(Propan-2-yl)oxy]propane | |
| Other names
Isopropyl ether
2-Isopropoxypropane Diisopropyl oxide DIPE | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1159 |
| |
| |
| Properties | |
| C6H14O | |
| Molar mass | 102.177 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Sharp, sweet, ether-like[1] |
| Density | 0.725 g/ml |
| Melting point | −60 °C (−76 °F; 213 K) |
| Boiling point | 68.5 °C (155.3 °F; 341.6 K) |
| 2 g/L at 20 °C | |
| Vapor pressure | 119 mmHg (20°C)[1] |
| -79.4·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H225, H316, H319, H335, H336, H361, H371, H412 | |
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P273, P280, P281, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P309+P311, P312, P332+P313, P337+P313, P370+P378, P403+P233, P403+P235, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | −28 °C (−18 °F; 245 K) |
| 443 °C (829 °F; 716 K) | |
| Explosive limits | 1.4–7.9% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
8470 mg/kg (rat, oral)[2] |
LDLo (lowest published)
|
5000-6500 mg/kg (rabbit, oral)[2] |
LC50 (median concentration)
|
38,138 ppm (rat) 30,840 ppm (rabbit) 28,486 ppm (rabbit)[2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 500 ppm (2100 mg/m3)[1] |
REL (Recommended)
|
TWA 500 ppm (2100 mg/m3)[1] |
IDLH (Immediate danger)
|
1400 ppm[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diisopropyl ether is secondary ether that is used as a solvent. It is a colorless liquid that is slightly soluble in water, but miscible with organic solvents. It is used as an extractant and an oxygenate gasoline additive. It is obtained industrially as a byproduct in the production of isopropanol by hydration of propylene.[3] Diisopropyl ether is sometimes represented by the abbreviation DIPE.
Uses
Whereas at 20 °C, diethyl ether will dissolve 1% by weight water, diisopropyl ether only dissolves 0.88%. Diisopropyl ether is used as a specialized solvent to remove or extract polar organic compounds from aqueous solutions, e.g. phenols, ethanol, acetic acid. It has also been used as an antiknock agent.
In the laboratory, diisopropyl ether is useful for recrystallizations because it has a wide liquid range.[4][5] Diisopropyl ether is used for converting bromoboranes, which are thermally labile, into isopropoxy derivatives.[6]
Safety
Diisopropyl ether forms explosive peroxides upon standing in air. This reaction proceeds more easily than for ethyl ether due to the increased lability of the C-H bond adjacent to oxygen. Many explosions have been known to occur during handling or processing of old diisopropyl ether.[7] Some laboratory procedures recommend use of freshly opened bottles.[4] Antioxidants can be used to prevent this process. The stored solvent are generally be tested for the presence of peroxides. It is recommended once every 3 months for diisopropyl ether compared to once every 12 months for ethyl ether.[8] Peroxides may be removed by shaking the ether with an aqueous solution of iron(II) sulfate or sodium metabisulfite.[9][10] For safety reasons, methyl tert-butyl ether is often used as an alternative solvent.
See also
- Dimethyl ether
- Diethyl ether
- Dipropyl ether
- Di-tert-butyl ether
- Methyl tert-butyl ether
- List of gasoline additives
References
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0362". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c "Isopropyl ether". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Sakuth, Michael; Mensing, Thomas; Schuler, Joachim; Heitmann, Wilhelm; Strehlke, Günther; Mayer (2010). "Ethers, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_023.pub2.
- ^ a b Andrea Goti, Francesca Cardona, Gianluca Soldaini (2005). "Methyltrioxorhenium Catalyzed Oxidation of Secondary Amines to Nitrones: N-Benzylidene-Benzylamine N-Oxide". Organic Syntheses. 81: 204. doi:10.15227/orgsyn.081.0204.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Ferenc Merényi, Martin Nilsson (1972). "2-Acetyl-1,3-Cyclopentanedione". Organic Syntheses. 52: 1. doi:10.15227/orgsyn.052.0001.
- ^ Shoji Hara, Akira Suzuk (1998). "Synthesis of 4-(2-Bromo-2-Propenyl)-4-Methyl-Y-Butyrolactone by the Reaction of Ethyl Levulinate with (2-Bromoallyl)Diisopropoxyborane Prepared by Haloboration of Allene". Organic Syntheses. 75: 129. doi:10.15227/orgsyn.075.0129.
- ^ Matyáš, Robert; Pachman, Jiří. (2013). Primary explosives. Berlin: Springer. p. 272. ISBN 978-3-642-28436-6. OCLC 832350093.
- ^ "Organic Peroxides - Hazards : OSH Answers". www.ccohs.ca. Canadian Centre for Occupational Health and Safety, Government of Canada.
- ^ Chai, Christina Li Lin; Armarego, W. L. F. (2003). Purification of laboratory chemicals. Oxford: Butterworth-Heinemann. p. 176. ISBN 978-0-7506-7571-0.
- ^ Hamstead, A. C. (1964). "Destroying Peroxides of Isopropyl Ether". Industrial and Engineering Chemistry. 56 (6): 37-42. doi:10.1021/ie50654a005.
External links
- CS1 maint: multiple names: authors list
- Articles without KEGG source
- Pages using collapsible list with both background and text-align in titlestyle
- Chembox having GHS data
- GHS warnings
- Articles containing unverified chemical infoboxes
- Articles with short description
- Dialkyl ethers
- Ether solvents
- Oxygenates
- Isopropyl compounds
- Symmetrical ethers
- Sweet-smelling chemicals
