Lactitol
 | |
| Clinical data | |
|---|---|
| Trade names | Importal, Pizensy, Lacty |
| Other names | Lactitol Hydrate (JAN JP) |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
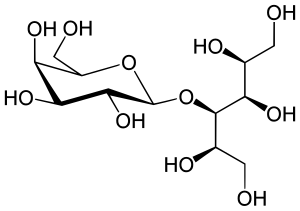
| Formula | C12H24O11 |
| Molar mass | 344.313 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 146 °C (295 °F) |
| |
| |
Lactitol is a sugar alcohol used as a replacement bulk sweetener for low calorie foods with 30–40% of the sweetness of sucrose. It is also used medically as a laxative. Lactitol is produced by two manufacturers, Danisco and Purac Biochem.[not verified in body]
Applications
Lactitol is used in a variety of low food energy or low fat foods. High stability makes it popular for baking. It is used in sugar-free candies, cookies (biscuits), chocolate, and ice cream, with a sweetness of 30–40% that of sucrose.[1] Lactitol also promotes colon health as a prebiotic. Because of poor absorption, lactitol only has 2–2.5 kilocalories (8.4–10.5 kilojoules) per gram,[1] compared to 4 kilocalories (17 kJ) per gram for typical saccharides. Hence, lactitol is about 60% as caloric as typical saccharides.
Medical
Lactitol is listed as an excipient in some prescription drugs.[2][3]
Lactitol is a laxative and is used to prevent or treat constipation,[4] e.g., under the trade name Importal.[5][6]
In February 2020, Lactitol was approved for use in the United States as an osmotic laxative for the treatment of chronic idiopathic constipation (CIC) in adults.[7][8][9]
Lactitol in combination with Ispaghula husk is an approved combination for idiopathic constipation as a laxative and is used to prevent or treat constipation.[medical citation needed]
Safety and health
Lactitol, erythritol, sorbitol, xylitol, mannitol, and maltitol are all sugar alcohols.[medical citation needed] The U.S. Food and Drug Administration (FDA) classifies sugar alcohols as "generally recognized as safe" (GRAS).[medical citation needed] They are approved as food additives, and are recognized as not contributing to tooth decay or causing increases in blood glucose.[medical citation needed] Lactitol is also approved for use in foods in most countries around the world.[medical citation needed]
Like other sugar alcohols, lactitol causes cramping, flatulence, and diarrhea in some individuals who consume it. This is because humans lack a suitable beta-galactosidase in the upper gastrointestinal (GI) tract, and a majority of ingested lactitol reaches the large intestine,[10] where it then becomes fermentable to gut microbes (prebiotic) and can pull water into the gut by osmosis.[medical citation needed] Those with health conditions should consult their GP or dietician prior to consumption.[medical citation needed]
History
The U.S. Food and Drug Administration (FDA) approved Pizensy based on evidence from a clinical trial (Trial 1/ NCT02819297) of 594 subjects with CIC conducted in the United States.[9] The FDA also considered other supportive evidence including data from Trial 2 (NCT02481947) which compared Pizensy to previously approved drug (lubiprostone) for CIC, and Trial 3 (NCT02819310) in which subjects used Pizensy for one year as well as data from published literature.[9]
The benefit and side effects of Pizensy were evaluated in a clinical trial (Trial 1) of 594 subjects with CIC.[9] In this trial, subjects received treatment with either Pizensy or placebo once daily for 6 months.[9] Neither the subjects nor the health care providers knew which treatment was being given until after the trials were completed.[9]
In the second trial (Trial 2) of three months duration, improvement in CSBMs was used to compare Pizensy to the drug lubiprostone which was previously approved for CIC.[9] The third trial (Trial 3) was used to collect the side effects in subjects treated with Pizensy for one year.[9]
References
- ^ a b Gränzle, M.G. (2011). "Lactose and Oligosaccharides | Lactose: Derivatives". Encyclopedia of Dairy Sciences (2nd ed.). Elsevier Ltd. pp. 202–208. doi:10.1016/B978-0-12-374407-4.00275-2. ISBN 9780123744074.
- ^ "Lactitol (Inactive Ingredient)". Drugs.com. 23 September 2018. Retrieved 24 February 2020.
- ^ "Lactitol Monohydrate (Inactive Ingredient)". Drugs.com. 3 October 2018. Retrieved 24 February 2020.
- ^ Miller LE, Tennilä J, Ouwehand AC (2014). "Efficacy and tolerance of lactitol supplementation for adult constipation: a systematic review and meta-analysis". Clin Exp Gastroenterol. 7: 241–8. doi:10.2147/CEG.S58952. PMC 4103919. PMID 25050074.
- ^ "Importal". Drugs.com. 3 February 2020. Retrieved 24 February 2020.
- ^ FASS.se (the Swedish Medicines Information Engine). Revised 2003-02-12.
- ^ "Pizensy: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 24 February 2020.
- ^ "Pizensy- lactitol powder, for solution". DailyMed. 21 February 2020. Retrieved 24 February 2020.
- ^ a b c d e f g h "Drug Trial Snapshot: Pizensy". U.S. Food and Drug Administration (FDA). 12 February 2020. Retrieved 4 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Grimble GK, Patil DH, Silk DB (1988). "Assimilation of lactitol, an unabsorbed disaccharide in the normal human colon". Gut. 29 (12): 1666–1671. doi:10.1136/gut.29.12.1666. PMC 1434111. PMID 3220306.
External links
 Media related to Lactitol at Wikimedia Commons
Media related to Lactitol at Wikimedia Commons- "Lactitol". Drug Information Portal. U.S. National Library of Medicine.
- Source attribution
- Articles with short description
- Use dmy dates from February 2020
- Infobox drug with local INN variant
- Articles containing unverified chemical infoboxes
- All articles with unsourced statements
- Articles with unsourced statements from March 2020
- Articles with unsourced statements from February 2020
- Commons category link is the pagename
- Disaccharides
- E-number additives
- Laxatives
- Sugar alcohols
- Sugar substitutes