Bis(triphenylphosphine)palladium chloride
| |
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C36H30Cl2P2Pd | |
| Molar mass | 701.90 g·mol−1 |
| Appearance | yellow powder |
| Melting point | 260°C (decomposed around 300°C) |
| Insoluble in water, acetone, ether, Carbon tetrachloride and n-heptane // Soluble in CHCl3 and CH2Cl2, Chloroform (Slightly), Methanol (Slightly, Heated) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 181.7°C |
| Related compounds | |
Related compounds
|
Bis(triphenylphosphine)platinum chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
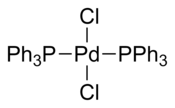
Bis(triphenylphosphine)palladium chloride is a coordination compound of palladium containing two triphenylphosphine and two chloride ligands. It is a yellow solid that is soluble in some organic solvents. It is used for palladium-catalyzed coupling reactions, e.g. the Sonogashira–Hagihara reaction. The complex is square planar. Many analogous complexes are known with different phosphine ligands.
Preparation and reactions
This compound may be prepared by treating palladium(II) chloride with triphenylphosphine:[1][2]
- PdCl2 + 2 PPh3 → PdCl2(PPh3)2
Upon reduction with hydrazine in the presence of excess triphenylphosphine, the complex is a precursor to tetrakis(triphenylphosphine)palladium, Pd(PPh3)4:[3]
- 2 PdCl2(PPh3)2 + 4 PPh3 + 5 N2H4 → 2 Pd(PPh3)4 + N2 + 4 N2H5+Cl−
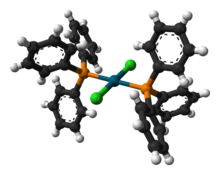
Structure
Several crystal structures containing PdCl2(PPh3)2 have been reported. In all of the structures, PdCl2(PPh3)2 adopts a square planar coordination geometry and the trans isomeric form.[4][5][6][7]
Applications
The complex is used as a pre-catalyst for a variety of coupling reactions.[8]
The Suzuki reaction was once limited by high levels of catalyst and the limited availability of boronic acids. Replacements for halides were also found, increasing the number of coupling partners for the halide or pseudohalide as well. Using bis(triphenylphosphine)palladium chloride as the catalyst, triflates and boronic acids have been coupled on an 80 kilogram scale in good yield.[9] The same catalyst is effective for the Sonogashira coupling.[10]
See also
References
- ^ Norio Miyaura; Akira Suzuki (1990). "Palladium-Catalyzed Reaction of 1-Alkenylboronates with Vinylic Halides: (1Z,3E)-1-Phenyl-1,3-octadiene". Organic Syntheses. 68: 130. doi:10.15227/orgsyn.068.0130.
- ^ Hiroshi Itatani, J.C.Bailar (1967). "Homogeneous Catalysis in the Reactions of olefinic Substances. V.Hydrogenation of Soybean Oil Methyl Ester with Triphenylphosphine and Triphenylarsine Palladium Catalysts". Journal of the American Oil Chemists' Society. 44: 147. doi:10.1007/BF02558176. S2CID 93580917.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ D. R. Coulson (1972). "Tetrakis(triphenylphosphine)palladium(0)". Inorganic Syntheses. Inorg. Synth. Inorganic Syntheses. Vol. 13. pp. 121–124. doi:10.1002/9780470132449.ch23. ISBN 9780470132449.
- ^ G. Ferguson, R. McCrindle, A. J. McAlees and M. Parvez (1982). "trans-Dichlorobis(triphenylphosphine)palladium(II)". Acta Crystallogr. B38 (10): 2679–2681. doi:10.1107/S0567740882009583.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ G. Steyl (2006). "trans-Dichlorobis(triphenylphosphine)palladium(II) dichloromethane solvate". Acta Crystallogr. E. 62 (6): m1324–m1325. doi:10.1107/S1600536806017521.
- ^ J. Pons, J. García-Antón, X. Solans, M. Font-Bardia, J. Ros (2008). "trans-Dichloridobis(triphenylphosphine)palladium(II)". Acta Crystallogr. E. 64 (Pt 5): m621. doi:10.1107/S1600536808008337. PMC 2961313. PMID 21202176.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ A. Naghipour, A. Ghorbani-Choghamarani, H. Babaee, M. Hashemi, B. Notash (2017). "Crystal structure of a novel polymorph of trans-dichlorobis (triphenylphosphine) palladium (II) and its application as a novel, efficient and retrievable catalyst for the amination of aryl halides and stille cross-coupling reactions". J. Organomet. Chem. 841: 31–38. doi:10.1016/j.jorganchem.2016.10.002.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ René Severin, Jessica Reimer, Sven Doye (2010). "One-Pot Procedure for the Synthesis of Unsymmetrical Diarylalkynes". J. Org. Chem. 75 (10): 3518–352. doi:10.1021/jo100460v. PMID 20420397.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ Jacks, T. E.; Belmont, Daniel T.; Briggs, Christopher A.; Horne, Nicole M.; Kanter, Gerald D.; Karrick, Greg L.; Krikke, James J.; McCabe, Richard J.; Mustakis; Nanninga, Thomas N. (2004). "Development of a Scalable Process for CI-1034, an Endothelin Antagonist". Organic Process Research & Development. 8 (2): 201–212. doi:10.1021/op034104g.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ^ Chinchilla, R.; Nájera, C. (2007). "The Sonogashira Reaction: A Booming Methodology in Synthetic Organic Chemistry". Chem. Rev. 107 (3): 874–922. doi:10.1021/cr050992x. PMID 17305399.
{{cite journal}}: CS1 maint: uses authors parameter (link)
- CS1 maint: uses authors parameter
- Chemical articles with multiple compound IDs
- Multiple chemicals in an infobox that need indexing
- Articles without EBI source
- Articles without KEGG source
- Articles without UNII source
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Palladium compounds
- Triphenylphosphine complexes
- Chlorides
- Chloro complexes

