Acetoacetyl-CoA
| |
| Names | |
|---|---|
| Preferred IUPAC name
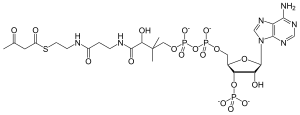
S-{(9R)-1-[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]-3,5,9-trihydroxy-8,8-dimethyl-3,5,10,14-tetraoxo-2,4,6-trioxa-11,15-diaza-3λ5,5λ5-diphosphaheptadecan-17-yl} 3-oxobutanethioate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| MeSH | acetoacetyl+CoA |
PubChem CID
|
|
| |
| |
| Properties | |
| C25H40N7O18P3S | |
| Molar mass | 851.609 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Acetoacetyl CoA is the precursor of HMG-CoA in the mevalonate pathway, which is essential for cholesterol biosynthesis. It also takes a similar role in the ketone bodies synthesis (ketogenesis) pathway of the liver. In the ketone bodies digestion pathway (in the tissue), it is no longer associated with having HMG-CoA as a product or as a reactant.
It is created from acetyl-CoA, a thioester, which reacts with the enolate of a second molecule of acetyl-CoA in a Claisen condensation reaction,[1] and it is acted upon by HMG-CoA synthase to form HMG-CoA. During the metabolism of leucine, this last reaction is reversed. Some individuals may experience Acetoacetyl-CoA deficiency.[2] This deficiency is classified as a disorder ketone body and isoleucine metabolism that can be inherited.[3]
Additionally, it reacts with NADPH-dependent acetoacetyl-coenzyme A reductase, also known as PhaB, in a pathway that produces polyester polyhydroxyalkanoate (PHA). The reduction of acetoacetyl-coA by Pha creates (R)-3-hydroxybutyryl-CoA, which polymerizes to PHA.[4] The pathway is present in bacteria such as Ralstonia eutropha and the PCC6803 strain of Synechocystis.[5]
References
- ^ Bruice PY (2017). Organic chemistry. Pearson. ISBN 978-0-13-404228-2. OCLC 974910578.
- ^ Tsuda H, Shiraki M, Inoue E, Saito T (August 2016). "Generation of poly-β-hydroxybutyrate from acetate in higher plants: Detection of acetoacetyl CoA reductase- and PHB synthase- activities in rice". Journal of Plant Physiology. 201: 9–16. doi:10.1016/j.jplph.2016.06.007. PMID 27372278.
- ^ Bose KS, Sarma RH (October 1975). "Delineation of the intimate details of the backbone conformation of pyridine nucleotide coenzymes in aqueous solution". Biochemical and Biophysical Research Communications. 66 (4): 1173–1179. doi:10.1016/0006-291x(75)90482-9. PMID 2.
- ^ Matsumoto K, Tanaka Y, Watanabe T, Motohashi R, Ikeda K, Tobitani K, et al. (October 2013). "Directed evolution and structural analysis of NADPH-dependent Acetoacetyl Coenzyme A (Acetoacetyl-CoA) reductase from Ralstonia eutropha reveals two mutations responsible for enhanced kinetics". Applied and Environmental Microbiology. 79 (19): 6134–6139. doi:10.1128/aem.01768-13. PMC 3811355. PMID 23913421.
- ^ Taroncher-Oldenburg G, Nishina K, Stephanopoulos G (October 2000). "Identification and analysis of the polyhydroxyalkanoate-specific beta-ketothiolase and acetoacetyl coenzyme A reductase genes in the cyanobacterium Synechocystis sp. strain PCC6803". Applied and Environmental Microbiology. 66 (10): 4440–4448. doi:10.1128/aem.66.10.4440-4448.2000. PMC 92322. PMID 11010896.
See also:
- Articles without KEGG source
- Articles without UNII source
- Articles with changed CASNo identifier
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Thioesters of coenzyme A
- All stub articles
- Biochemistry stubs
